Valve Surgery
The heart has four valves, the tricuspid, pulmonary, mitral, and aortic. Heart valves are designed to direct the flow of blood through the heart in one direction. Blood coming from the upper and lower extremities enters the heart through the right atrium. It then goes through the tricuspid valve into the right ventricle. From the right ventricle it is pumped through the pulmonary valve into the lungs. The carbon dioxide is removed and the blood is oxygenated in the lungs. Oxygenated blood from the right and left lungs then collects in the left atrium. It then passes through the mitral valve into the left ventricle. Blood is pumped out of the left ventricle through the aortic valve to the entire body.Various diseases can affect heart valves. Heart valves can be affected in one of three ways:
- Stenosis or narrowing of the valve opening. In this process the valve cannot fully open to allow blood to leave from one chamber to the next. When valve stenosis occurs blood has to be more forcefully pushed out of from one chamber to the next. This usually results in the one chamber enlarging, and increasing in muscle mass.
- Regurgitation or insufficiency is when the valve is unable to close properly. This allows blood to leak back from one chamber to the previous chamber. The overload of blood can cause that chamber to enlarge to accommodate the excess blood.
- Combination of stenosis and regurgitation. In this condition the valve can neither open nor close effectively. Thus blood has to be forcefully pushed out and then some of it will leak back.
Aortic Valve Disease
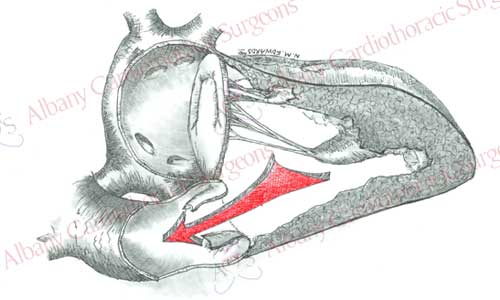
The aortic valve is composed of three crescent shaped leaflets, which are attached to a rim of tissue (annulus) between the aorta and the left ventricle. Normally the valve opens with each heartbeat, allowing blood to leave the left ventricle and go to rest of the body. As the heart beat ends, the valve closes and preventing blood from returning to the heart. The leaflets are thin and pliable, however with disease this anatomy can change. The leaflets may narrow, obstructing the flow of blood out of the heart, they may not close properly allowing blood to flow back into the heart, or a combination of these two. All these conditions cause the heart to work harder and increase in size. Eventually the heart is unable to keep up with the increased demands and begins to fail. Patients may experience this as shortness of breath, lightheadedness, or chest discomfort. Valve replacement can help reverse this process, decrease symptoms, and increase longevity.
The aortic valve can be damage by various diseases. A degenerative process due to aging, infection, rheumatic fever, and congenital defects are the most common causes. One percent of the population is born with a valve with two leaflets (bicuspid) rather than three (tricuspid). Most of these valves develop changes by the fifth or sixth decade, which may require replacement.
Aortic valve replacement is accomplished with a prosthetic valve. Two types of prosthetic valves are utilized. A mechanical valve is made of inert non-biologic tissue. It has a low profile design and can last for many years without structural deterioration. A disadvantage is the lifelong requirement for anticoagulation (blood thinners). Biologic valves (bioprosthetic) are made of specially treated biologic tissue, which makes them immunologically inert. Biologic valves have the advantage that anticoagulation is not needed. However the biologic tissue may deteriorate (over 15-18 years) with time causing the valve to fail. For this reason biologic valves are most commonly used in individuals over the age of 65.
 St. Jude Medical Mechanical Valve
St. Jude Medical Mechanical Valve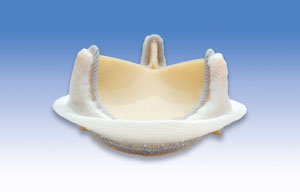 Edwards Bovine Bioprosthetic Pericardial Valve
Edwards Bovine Bioprosthetic Pericardial ValveSurgical Technique for Aortic Valve Replacement
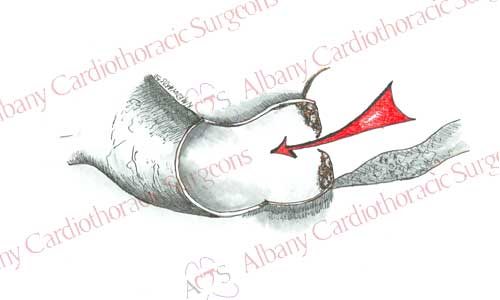
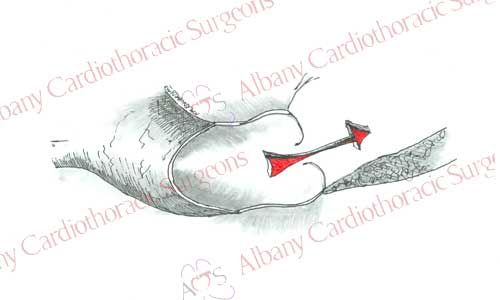
Aortic valve replacement requires the use of a general anesthetic. Patients are placed on the heart lung machine by placing catheters in the aorta and right atrium. The heart-lung machine does the work of the heart and lungs so that blood can be redirected from the heart. Once the patient is placed on the heart lung machine a clamp is placed on the aorta to stop blood flow into the heart. This allows surgeons to work on a motionless heart devoid of blood. The aortic valve is exposed by opening the ascending aorta. The diseased valve leaflets are removed. Sutures with Teflon felt bolsters (pledgets) are placed along the rim of tissue where the native valve was once attached. Sutures are then passed through the prosthetic valve. Prosthetic valve is then secured in place by tying the sutures. The aorta is then closed and the clamp removed from the aorta. This allows blood flow to resume to the heart and the heart begins to beat. Any air is evacuated from the heart; the support of the heart lung machine is withdrawn. Temporary pacing wires are placed, drainage tubes inserted, and the incisions are closed. The patient is then taken to the intensive care unit for recovery.
Mitral Valve Disease
The mitral valve is a complex structure between the left atrium and left ventricle. It is the second most common valve requiring surgical intervention. The valve is composed of two leaflets attached to fibrous tissue in the heart. The leaflets also have fine chords and muscular attachments to the left ventricle. In addition to valve function these structures can affect left ventricular function and shape. Blood coming from the lungs collects in the left atrium, passes through the valve into the left ventricle. Normal functioning of the valve prevents blood from going back into the left atrium when the left ventricle contracts.
Various diseases can damage the mitral valve causing stenosis, regurgitation, or both. Rheumatic fever and degenerative changes are the most common causes of mitral stenosis. Valve leaflets become thick, less pliable, resulting in stenosis. In order to force blood through the narrowed orifice, the left atrium increases in size and becomes more muscular. Eventually the high pressures generated in the left atrium will be transmitted to the lungs, resulting in shortness of breath. Degenerative changes, infection, heart attack and rheumatic fever are the most common causes of mitral regurgitation. With each beat a quantity of blood leaks back into the left atrium. The increased volume of blood in this chamber eventually causes it to enlarge. With time there will also be a larger volume of blood ejected into the left ventricle, causing this chamber to dilate. If the process is allowed to continue it will result in shortness of breath and heart failure. Repair or replacement of the valve can improve symptoms, longevity, and heart muscle function.
The mitral valve can be replaced with a prosthetic valve. Two types of prosthetic valves are utilized. A mechanical valve is made of inert non-biologic tissue. It has a low profile design and can last for many years without structural deterioration. A disadvantage is the lifelong requirement for anticoagulation (blood thinners). Biologic valves (bioprosthetic) are made of specially treated biologic tissue, which makes them immunologically inert. Biologic valves have the advantage that anticoagulation is not needed. However the biologic tissue may deteriorate (over 12-15 years) with time causing the valve to fail. For this reason biologic valves are most commonly used in individuals over the age of 65.
The mitral valve can also be repaired. Surgeons have developed a variety or techniques to repair the valve based on the anatomic changes causing regurgitation. These techniques often involve removing a damaged portion of the valve leaflet, followed by reconstruction of the remaining valve. Placing a prosthetic ring around the valve completes the valve repair. The ring supports the repair and prevents further dilatation of the valve. Valve repair has the advantage of preserving the native valve and its anatomic attachments to the left ventricle. This results in better long term function of the left ventricle. Long term anticoagulation is also not required with valve repair decreasing the risk of bleeding complications. Thus when possible valve repair is preferable to valve replacement.
Surgical Technique for Mitral Valve Repair/Replacement
Depending on the structural changes that occur, the mitral valve can be either replaced or repaired. The procedure is performed using general anesthesia. Once the patient is asleep, the heart is exposed. Patients are placed on the heart lung machine by placing catheters in the aorta and right atrium. The heart-lung machine does the work of the heart and lungs so that blood can be redirected from the heart. Once the patient is placed on the heart lung machine a clamp is placed on the aorta to stop blood flow into the heart. This allows surgeons to work on a motionless heart devoid of blood. The mitral valve is most commonly exposed through the left atrium. Once exposed the valve and its supporting structures are carefully examined to determine if the valve can be repaired or replaced.
If the valve is to be replaced, the leaflets are excised. Depending on the extent of damage to the valve attempts are made to preserve either one of the leaflets and/or the fine chordal attachments of the valve. Preservation of these structures helps maintains better function of the left ventricle. Sutures with Teflon felt bolsters (pledgets) are passed through the rim of tissue left after valve excision and through the new valve. Sutures are then tied securing the valve in place. Depending on the patient’s age either a mechanical or bioprosthetic valve can be used.
If the valve is to be repaired, this is completed and a ring is sutured around the valve. After the repair is completed, competency of the valve is tested. This is accomplished by passively filling the left ventricle and observing for any leakage through the valve.
Once the valve is replaced or repaired, the left atrium is closed and the clamp removed from the aorta. This allows blood flow to resume to the heart and it begins to beat. Air is evacuated from the heart; support of the heart lung machine is withdrawn. The valve repair is once again reviewed using echocardiography to ensure a satisfactory repair. Temporary pacing wires and drainage tubes are placed and the incisions are closed. The patient is taken to the intensive care unit for recovery.
Minimally Invasive Heart Valve Surgery
The goal of minimally invasive valve surgery is to approach the valves through smaller incisions. This approach offers better cosmesis than the standard midline incision, while maintaining the quality of the valve surgery. Minimally invasive incisions are often 6-8 cm (3-4 in) in length and they maybe on the side of the chest (thoracotomy or Heartport) or in the midline (partial sternotomy). In the thoracotomy incision the chest is entered between the ribs. It provides good access to the valves, however alternative methods have to be used to place the patient on the heart lung machine. This is most commonly is done by using blood vessels in the groin (femoral artery and vein). In the partial sternotomy incision, only the upper part of the breast bone is split. Access for the heart-lung machine can often be done using blood vessels in the chest when this incsion is used.
Although the major advantage of a minimally invasive incision is cosmetic, other advantages may include:
- Less pain
- Less blood loss
- Less risk of bone infection
- Faster recovery
It is important to note that not all patients are candidates for minimally invasive valve surgery. Patients with obesity, concomitant coronary artery disease, or severe atherosclerosis in groin and abdominal blood vessels are not good candidates.
Tricuspid Valve Surgery
The tricuspid valve prevents reflux of blood from the right ventricle to the right atrium. Tricuspid insufficiency can lead to swelling (edema) of the extremities and fatigue. Our surgeons are experienced in tricuspid valve repair and replacement.